
The safety and efficacy for the treatment of patients who have back pain as the greatest region of pain have not been evaluated.Įxternal defibrillators. The safety and effectiveness of neurostimulation for pediatric use have not been established.īack pain. Safety and effectiveness of neurostimulation for use during pregnancy and nursing have not been established. Implanting physicians should be experienced in the diagnosis and treatment of chronic pain syndromes and have undergone surgical and device implantation training for dorsal root ganglion (DRG) neurostimulation systems. 
The following warnings apply to this neurostimulation system.Ĭlinician training. Some models of this system are Magnetic Resonance (MR) Conditional, and patients with these devices may be scanned safely with magnetic resonance imaging (MRI) when the conditions for safe scanning are met.įor more information about MR Conditional neurostimulation components and systems, including equipment settings, scanning procedures, and a complete listing of conditionally approved components, refer to the MRI procedures clinician's manual for neurostimulation systems (available online at medical.abbott/manuals).įor more information about MR Conditional products, visit the Abbott Medical product information page at neuromodulation.abbott/us/en/healthcare-professionals/mri-support.html. Patients who failed to receive effective pain relief during trial stimulation are contraindicated to process to the permanent implant procedure. This neurostimulation system is contraindicated for patients who are Changes secondary to neuropathic pain seen in CRPS I (RSD) may be present, but are not a diagnostic requirement for CRPS II (causalgia). Nerve damage may result from traumatic or surgical nerve injury. CRPS II (causalgia) is defined as a painful condition arising from damage to a nerve. **Please note that in 1994, a consensus group of pain medicine experts gathered by the International Association for the Study of Pain (IASP) reviewed diagnostic criteria and agreed to rename reflex sympathetic dystrophy (RSD) and causalgia as complex regional pain syndrome (CRPS) types I and II, respectively. *Study subjects from the ACCURATE clinical study had failed to achieve adequate pain relief from at least two prior pharmacologic treatments from at least two different drug classes and continued their pharmacologic therapy during the clinical study. This neurostimulation system is indicated for spinal column stimulation via epidural and intraspinal lead access to the dorsal root ganglion as an aid in the management of moderate to severe chronic intractable* pain of the lower limbs in adult patients with complex regional pain syndrome (CRPS) types I and II.** The system is intended to be used with leads and associated extensions that are compatible with the system. This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. For specific indications, contraindications, instructions, warnings, precautions, and adverse effects about system components available in your country or region, see the approved clinician's manual for those components. Read this section to gather important prescription and safety information. 2015 18(1):50-7.ĭorsal Root Ganglion Therapy (DRG) Therapy Prescription And Safety Information Lack of body positional effects on paresthesias when stimulating the dorsal root ganglion (DRG) in the treatment of chronic pain. Unique characteristics of the dorsal root ganglion as a target for neuromodulation. Esposito M, Malayil R, Hanes M, Deer T.Dorsal root ganglion stimulation yielded higher treatment success rate for complex regional pain syndrome and causalgia at 3 and 12 months: a randomized comparative trial. Proclaim™ DRG Neurostimulation System Clinician’s Manual. 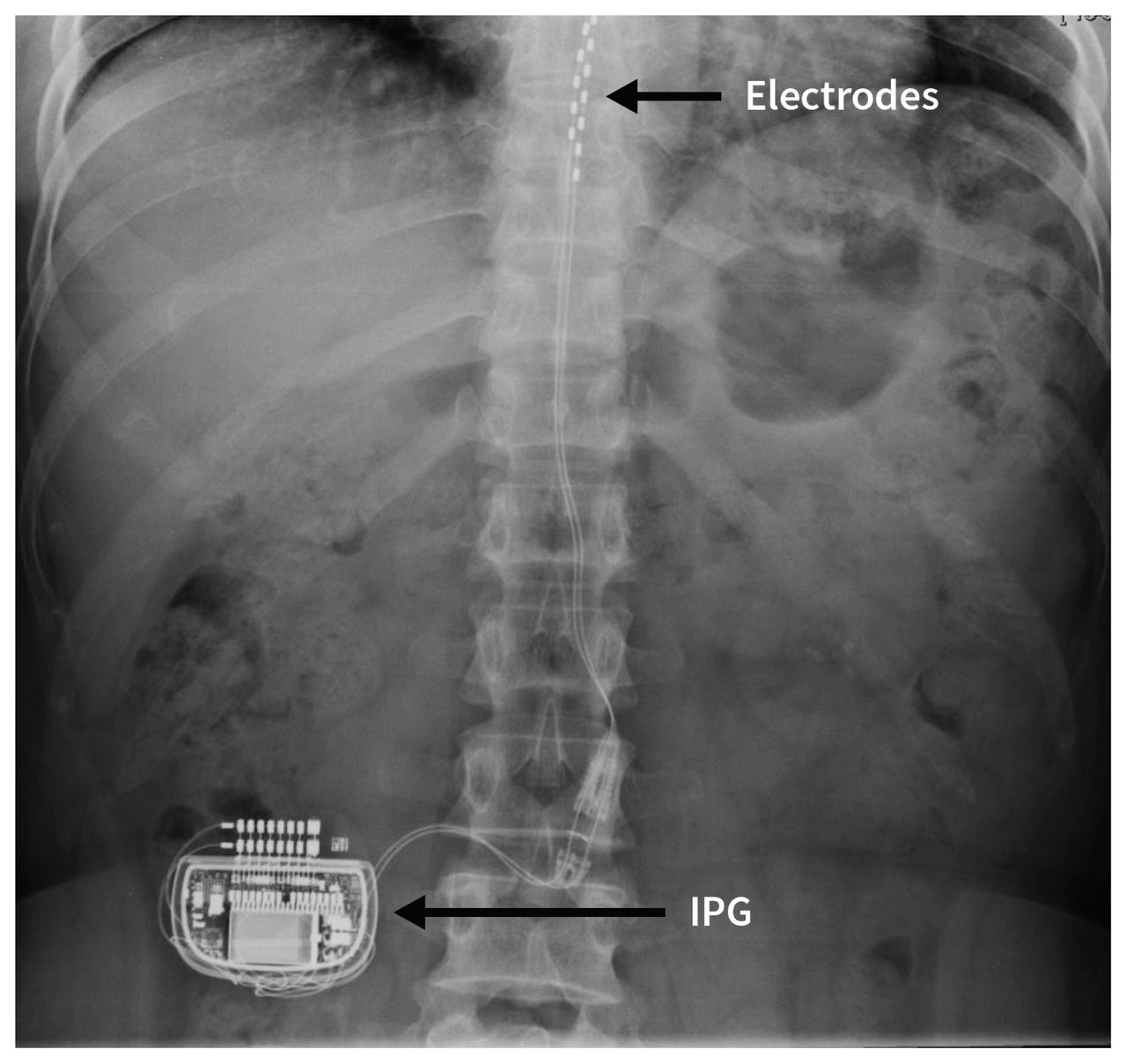
For a list of mobile devices compatible with Abbott Neuromodulation Patient Controller applications, visit. § Dual-lead system with one-year shelf life at 1600-ohms impedance and 24 hours of 20-Hz frequency, 300-µs pulse width, and 0.8-mA amplitude stimulation. † When compared to traditional tonic spinal cord stimulation based on outcomes from the ACCURATE investigational device exemption study. * Within approved parameters. Refer to the Instructions for Use for full details on the MR Conditional scan parameters. ‡ Indicates a third party trademark, which is property of its respective owner. 
™ Indicates a trademark of the Abbott group of companies.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
